פוטנציאל ריפוי עם Axicabtagene Ciloleucel (Axi-Cel) , הערכת שרידות לטווח ארוך בחולים בלימפומה נשנית של תאי B גדולים במחקר Zuma-1, כנס ASH 2023
At the latest ASH conference 2023, Insights from exploratory analyses in Phase 2 of ZUMA-1 presented, utilizing proposed endpoints for assessing potential cures with survival analyses extending up to a 6-year follow-up period.
Background:
Extended outcomes from ZUMA-1, a Phase 1/2 study involving multiple centers and a single-arm design, for patients with refractory Large B-Cell lymphoma, revealed persistent overall survival (OS) rates, indicating a median of 25.8 months and a 5-year estimate of 43%.1
Initial assessments of disease-specific survival (DSS) in ZUMA-1 suggested Axicabtagene ciloleucel may be curative for a subset of patients. Nevertheless, there is currently no clear establishment of endpoints that define the potential for a cure in patients with LBCL. Here, we report exploratory analyses from the Phase 2 portion of ZUMA-1 using proposed endpoints to measure cure along with survival analyses with up to 6 years of follow-up.
Lymphoma-related event-free survival (LREFS) was defined as the time from axi-cel infusion until disease progression, initiation of new lymphoma therapy, autologous stem cell transplantation, or death due to progression, whichever was earliest. DSS was defined as time from axi-cel infusion to death due to progression or axi-cel–related adverse events (AEs).
Results:
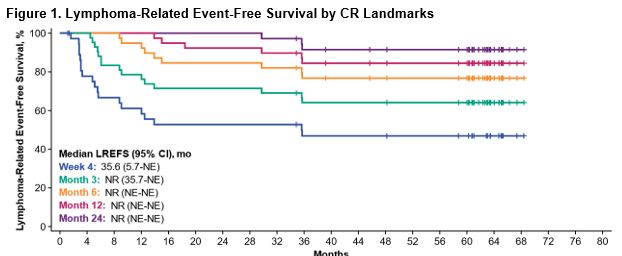
At a median follow-up of 63.1 months in the 5-year analysis (data cutoff on August 11, 2021), median LREFS was 5.8 months (95% CI, 3.4-13.9) and the 60-month LREFS rate by KM estimate was 33.5%. Among patients who achieved a complete response (CR) as best response (n=59), median DOCR was not reached (95% CI, 12.9 months-not estimable).
Median LREFS among those with a CR was not reached (95% CI, 18.4 months-not estimable) and the 60-month LREFS rate was 56.8%.
Among those with a CR at Months 12 and 24, 60-month estimated rates of LREFS and ongoing CR were >80% (84.5% and 91.5%, respectively; Figure 1).
The data cutoff date for the 6-year survival analysis was August 11, 2022. Median OS was 25.8 months (95% CI, 12.8-63.7). In a competing risk assessment of OS, the cumulative incidence of death at 72 months was 61.6%.
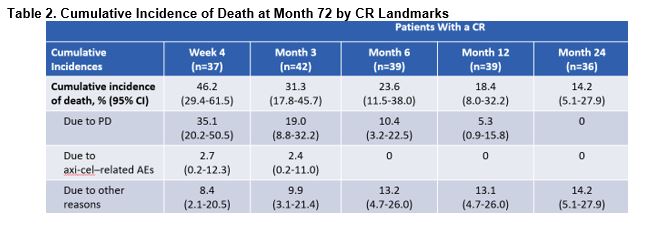
Between Months 24 and 72, there appeared to be an increase of 6% in the cumulative incidences of death due to progression (40.6% and 46.6%, respectively) and other reasons (5.0% and 11%, respectively), while deaths due to axi-cel–related AEs remained 4.0% at each timepoint.
Those who had a CR at Months 12 and 24 had a 72-month cumulative incidence of death of 18.4% and 14.2%, respectively (Table 2). Additionally, those who had a CR at 12 and 24 months had 72-month KM-estimated DSS of 94.4% and 100%, respectively.
 Conclusion:
Conclusion:
In this post hoc analysis of ZUMA-1 with up to 6 years of follow-up, axi-cel exhibited prolonged long-term durable responses in a significant proportion of patients, with a 5-year rate reaching 33.5% (56.8% for those who attained a complete response). Moreover, achieving a complete response at 12 and 24 months may serve as a potential predictor for extended overall survival. Notably, among patients who achieved a complete response, deaths attributed to axi-cel–related adverse events primarily occurred before Month 24. These results demonstrate the curative potential of axi-cel in a subset of patients with R/R LBCL.3
- Neelapu SS, et al. 2023
- Neelapu et al. N Engl J Med. 2017
- Neelapu et al. ASH 2023 Abstract #4864
Yescarta is indicated for the treatment of adult patients with diffuse large B-cell lymphoma (DLBCL) and high-grade B-cell lymphoma (HGBL) that relapses within 12 months from completion of, or is refractory to, first-line chemoimmunotherapy.
Yescarta is indicated for the treatment of adult patients with relapsed or refractory (r/r) diffuse large B-cell lymphoma (DLBCL) and primary mediastinal large B-cell lymphoma (PMBCL), after two or more lines of systemic therapy.
Limitation of Use: Yescarta is not indicated for the treatment of patients with primary or secondary central nervous system lymphoma.
Yescarta is indicated for the treatment of adult patients with relapsed or refractory follicular lymphoma (FL) after two or more lines of systemic therapy.
For further information and full list of adverse events, please refer to the approved prescribing information as available at Israeli Ministry of Health website:
https://israeldrugs.health.gov.il/#!/byDrug
Any suspected adverse events should be reported to the Ministry of Health according to the National Regulation by using an online form at https://sideeffects.health.gov.il/
Additionally, adverse events can be reported to Gilead: Safety_FC@gilead.com
This article is intended for healthcare providers only.
For any further questions please contact: medinfo.israel@gilead.com
© Gilead Sciences Israel Ltd. All rights reserved.
IL-YES-0151, FEB 2024



